|
Hg behaves as a transition metal atom and transfers electrons from its closed-shell 5d orbitals to F atoms in Hg–F compounds at high pressure 12. The completely filled 5p inner shell of Cs can be activated to form unexpected F-rich Cs–F compounds, causing Cs to expand beyond the +1 oxidation state 11. Furthermore, previous studies have indicated that fluorine, the most electronegative halogen 10, can activate the closed shell of an element in fluoride at high pressure. Two fascinating compounds, Na 3Cl and NaCl 3, with unusual compositions that violate the octet rule were predicted in theory and confirmed by high-pressure experiments 4. At high pressure, the classical chemistry rules regarding halogens are further broken. Interestingly, halogens also exhibit hypervalent features in their compounds, such as XeF 2 and CsI 3, under moderate conditions 8, 9. Group VIIA elements (halogens) with partially filled p shells are usually stable in a −1 charge state at ambient pressure, satisfying the octet rule of chemistry, wherein table salt, NaCl, is the most well-known compound, and its chemistry is well understood. The noble gas Xe, a chemically inert element with a completely filled shell under ambient conditions, captures electrons under high pressure, becoming negatively charged in Mg–Xe compounds 7. 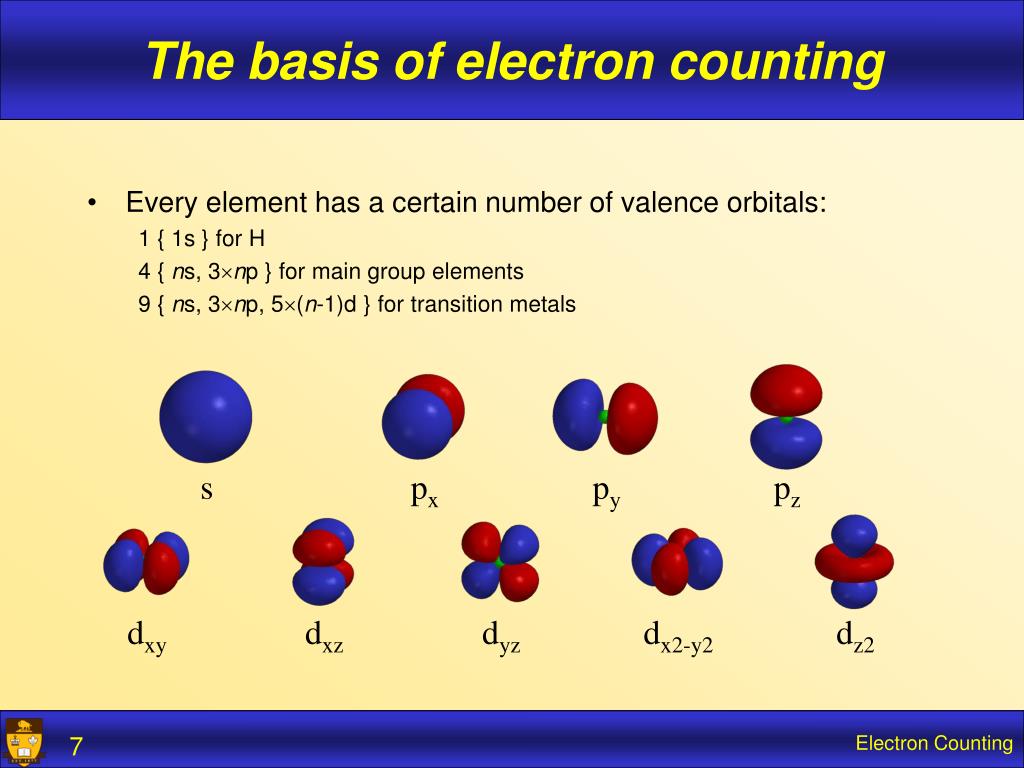
For example, the most “simple” metal, Na, undergoes an unusual metal–insulator phase transition at high pressure due to unexpected p–d orbital hybridizations 6. At high pressure, the electronic configuration of an atom will change drastically, enabling matter to exist in a totally counterintuitive chemical regime. However, in a high-pressure environment, the rules of classical chemistry are broken 4, 5. An element with a completely filled outer electron shell is highly stable under ambient conditions. 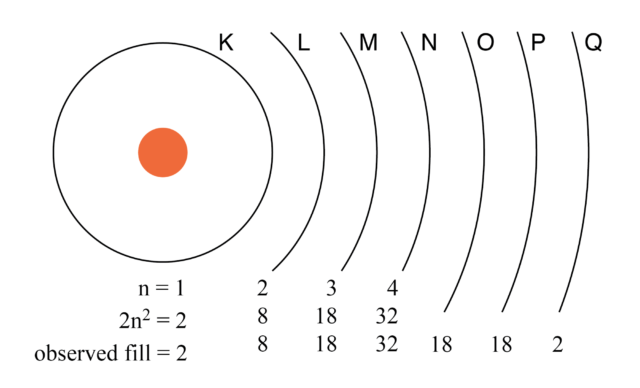
Generally, to satisfy the octet rule, an element gains or loses electrons to fulfill its outer electron shell, attaining the electronic configuration of the nearest noble gas element 2, 3. The fundamental chemical properties of a given element strongly depend on its electronic configuration 1.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
